AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
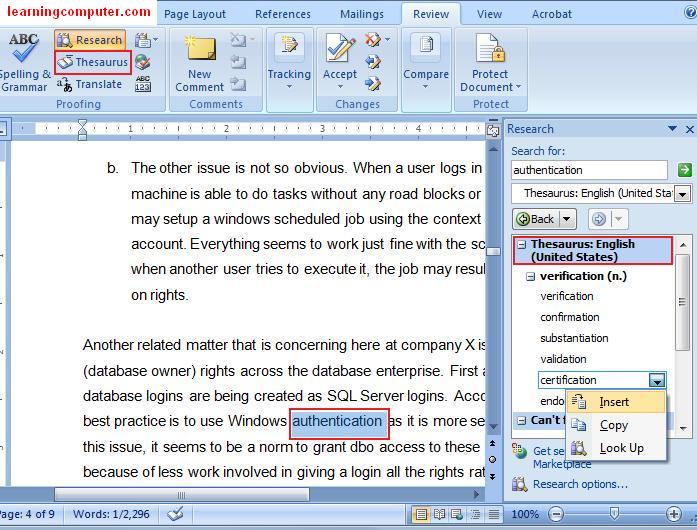
 Data included in research reports must be original and should be as timely and current as possible (see Timeliness of Data). Each manuscript should clearly state an objective or hypothesis the design and methods (including the study type and setting and dates, patients or participants with inclusion and exclusion criteria and/or participation or response rates, or data sources, and how these were selected for the study) the essential features of any interventions the main outcome measures the main results of the study a discussion section placing the results in context with the published literature and addressing study limitations and the conclusions and relevant implications for clinical practice or health policy. 
These reports typically include randomized trials (see Clinical Trial), intervention studies, cohort studies, case-control studies, epidemiologic assessments, and other observational studies. Decisions and Management of Editorial Conflicts of Interest.Personal Communications and Unpublished Data.Ethical Approval of Studies and Informed Consent.Reporting Demographic Information for Study Participants.Data Access, Responsibility, and Analysis. 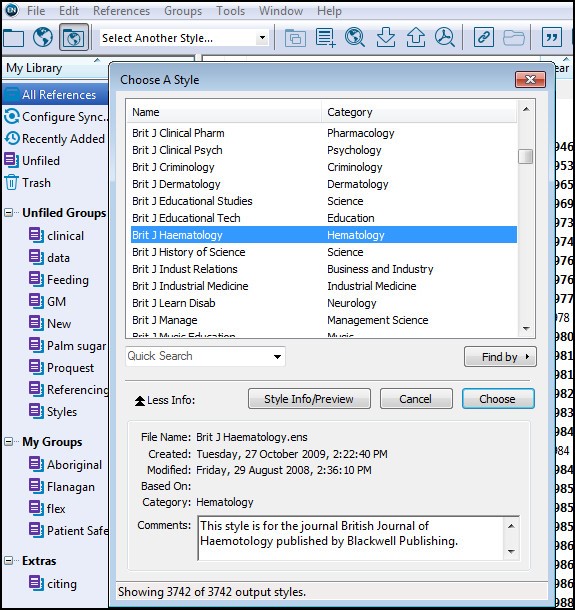
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed